Kanvas Biosciences
Mapping the microbiome to improve human health
The microbiome is the last unmapped territory in the human body, home to around 39 trillion cells, or about 43 percent of all the cells in or on our bodies. We know these bacteria and other microbial organisms teem in large colonies in our guts, mouths, lungs, and on our skin. But we don’t know exactly where they are or what they’re doing. That matters, because if we can’t characterize how these microbes interact with each other and with our own cells, we don’t really know how they affect health and disease.
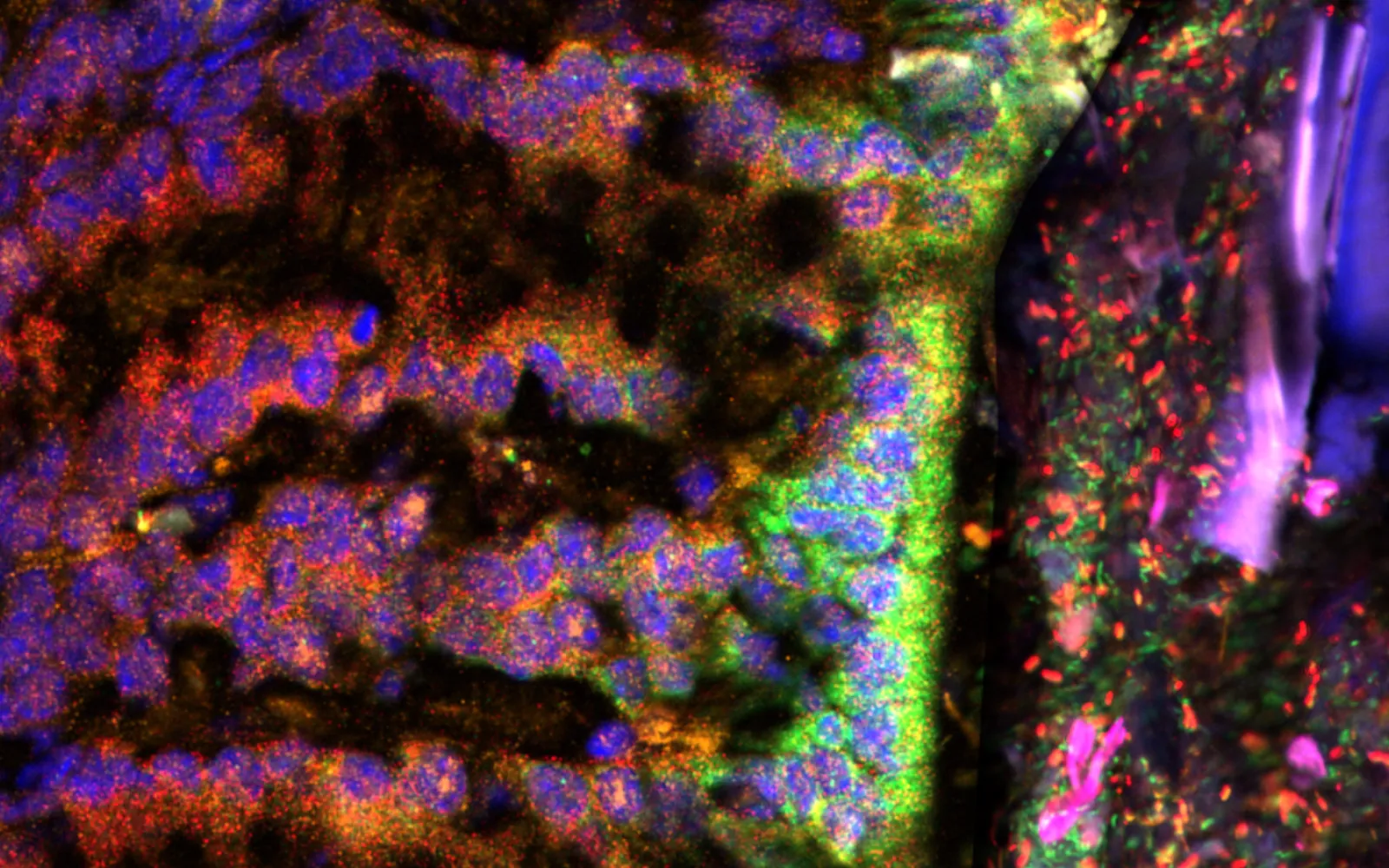
A new DCVC portfolio company, Kanvas Biosciences, will change all that. Kanvas has invented an imaging technology that reveals, with stunning resolution, the full range of messenger RNA transcripts made by both microbial communities and host cells. This ability to map the molecules that direct protein synthesis reveals not just which microbes are present in the microbiome but which genes they’re actively expressing, which genes the human cells are expressing, and how they are affecting each other — providing a detailed picture of host-microbiome interactions in the gastrointestinal tract or anywhere else microbes thrive in the body.
Kanvas has drawn, for the first time, a living atlas of the entire dance of microbial and human cells. If we hope to understand how disorders of the microbiome — called dysbioses — make people ill, or how antibiotic resistance develops and can be inhibited, or how we might drug the microbiome as if it were any other organ in the body, we’ll need that level of resolution. That’s why DCVC is leading a $12 funding round for the company, announced today.
Toward making the microbiome a druggable organ
Because microbes are the descendants of species that co-evolved with us over millions of years, we couldn’t do without them: they drive a multitude of functions including digestion, immune system regulation, neurological processes, even infant development. The microbiome is involved in many diseases, not all of them in the gut, including cancer, diabetes, and cardiovascular and neurological disorders. Obese people suffer from an imbalance in the gut’s microbial community; so do many children with autism. The microbiome even metabolizes pharmaceuticals and modulates treatment response. But while we have known humans are multi-organismal creatures since the 19th century, these tantalizing insights into the microbiome have often failed to deliver new medicines.
DCVC believes that Kanvas’s new spatial biology capability, which combines machine learning with an ingenious binary barcoding system invented by the company, will speed the diagnosis of microbial diseases and the development of biotherapeutic products. DCVC was drawn to the company because its founders think like explorers. Matthew Cheng, Kanvas’s co-founder and CEO, says he sees the microbiome as literally uncharted frontier — but one that, given the right tools, can be visualized at cellular resolution even as it’s characterized on a functional, genetic level. He explains that Kanvas’s barcoding system, which labels RNA targets with pairs of glowing molecules called fluorophores, is designed to show not only the identities and functions of the microbes in a given location, but also how local host cells are responding. “We think that having these two additional axes of information will be the difference between understanding that the microbiome is relevant to human health and actually characterizing it as a druggable organ system,” Cheng says.
Kanvas’s founders first described their unique technology in a paper published in Nature in 2020. It’s called HiPR-FISH — short for “High Phylogenetic Resolution microbiome mapping by Fluorescence In Situ Hybridization” — and the first step in the process is to label cells with nucleic acid probes customized to bind to the RNA of specific taxa of bacteria or other microbes. Each probe bears a pair of readout sequences. In the second step, these end sequences bind to fluorophores, which show distinct spectral peaks under different wavelengths of laser light. Each probe ends up sporting two fluorophores — the barcode for that taxon. By scanning the labeled cells on a confocal microscope, Kanvas’s system collects a mix of signals, which machine learning algorithms sort back into individual barcodes. The method is highly multiplexed, meaning that a large number of barcodes can be distinguished simultaneously (2n‑1 barcodes for each imaging round, where n is the number of fluorophores used; with just 10 fluorophores the system can identify 1,023 different RNA transcripts).
Getting started
No other company can map the microbiome and determine cellular function in such detail. Kanvas’s imaging system will become an indispensable tool for any health or biomedical organization wishing to profile the microbiome, evaluate host-microbiome interactions, or develop living biotherapeutic products. In one experiment, for example, Kanvas was able to show that a specific cocktail of beneficial bacteria can protect against an overpopulation of Gardnerella vaginalis, a type of bacteria that can cause vaginosis. A dramatic micrograph shows the cocktail adhering preferentially to vaginal epithelial cells, virtually eliminating Gardnerella from the sample. “Because of our ability to look not only at what’s happening in the microbiome, but also in real time see what’s happening in the host,” Cheng says, “we can design the next generation of microbiome therapeutics: studying engraftment, safety, and efficacy all in the same assay.”
Lions Capital LLC, Cooke LLC, Uncommon Denominator, and Triple Impact Capital partnered with DCVC to assemble this round of funding. As part of the investment round, I’m pleased to be joining Kanvas’s board of directors. DCVC also invested in Kanvas’s seed round of financing in 2021, and we can’t wait to see the speculations of the field confirmed or denied, and for drug discovery to gain a new territory to explore.
Jason Pontin is a General Partner at DCVC and a cofounder of DCVC Bio portfolio company Totus Medicines.